Pharmaceutical-technology.com – November 2011
Although pharmaceutical companies rely heavily on contractors, the industry is still new to the world of outsourcing. Risk management strategies across parts of the pharmaceutical supply chain remain surprisingly unsophisticated. Elly Earls finds out why openness, transparency and up-to-date risk management tools are essential for ensuring pharmaceutical companies avoid unnecessary risks and take advantage of unexpected opportunities.
Both Big Pharma and their smaller biotech counterparts are increasingly relying on the services of outside contractors for specialist operations such as sterile manufacturing or the running of clinical trials, a strategy that allows the sponsor company to focus on its core competences. But while this practice has been commonplace in industries like defence and aerospace for decades, the pharmaceutical industry is still relatively new to such a collaborative way of working.
“Both Big Pharma and their smaller biotech counterparts are increasingly relying on the services of outside contractors.”
“Pharma has been rather late to the party, a lot of the big companies have been very used to being self-sufficient,” said John Bennett, owner and director of JABPharma Ltd, an independent pharmaceutical consultancy specialising in project, programme, portfolio and risk management. “But the use of contract research has doubled over the last 15 years and I believe this trend will only continue to grow as more and more companies get rid of their internal capabilities.”
The increased use of outsourced clinical trial support has been supported by the expanding capabilities of contract research organisations (CROs).
“15 years ago, there were very few, if any, companies that were capable of running a global trial, but now it’s very much the opposite,” Bennett noted. “On top of this, larger pharmaceutical companies see using CROs as a way of providing a much more flexible workforce, which is critical in the dynamic pharmaceutical environment.”
For Kristie Zinselmeier, senior director of marketing at BioPharma Solutions, a business unit of the Baxter Healthcare Corporation, which partners with pharmaceutical companies to provide sterile manufacturing solutions, it’s a similar story in the contract manufacturing arena.
“We’ve seen a steady increase over the past ten years,” she confirmed. “We’re seeing an increased recognition that scarce resources need to be concentrated in the areas which drive the most value for Pharma.”
Risky business
Yet, while outsourcing brings with it a whole range of benefits, there are inevitable risks associated with allowing another organisation to run a significant part of your project. “Loss of control is the major risk,” Bennett said. “It’s important to bear in mind that, irrespective of how a clinical trial is performed, the sponsor is ultimately accountable for it.”
“Where it gets slightly trickier, however, is ensuring communication between the two parties is maintained.”
Assessing the financial stability of a potential provider is therefore an absolutely critical parameter in risk mitigation strategies, when it comes to both contract research and contract manufacturing.
“In the past year we’ve seen CMOs exit the market and this has created stress in the pharmaceutical supply chain,”Zinselmeier said. “You need to not only understand business risk as it relates to the service provider’s solvency, but also as it relates to sustaining manufacturing investment to maintain compliance.”
In Bennett’s view, large pharmaceutical companies are generally very diligent at ensuring financial and legal risks of this type are minimised. “They will pay a lot of attention to making sure their CROs have all the right operating procedures in place and use all the right quality checks,” he said.
Where it gets slightly trickier, however, is ensuring communication between the two parties is maintained. “What we’ve heard from CROs is that sponsors often don’t want to hear about the operational risks; they want CROs to be incredibly optimistic about timelines – to the point where they become very difficult to achieve,” Bennett explained.
“The maturity of the dialogue between the sponsor and the CRO isn’t as it might be in other industries. There is a communication barrier and risk is seen as a negative.”
Fiona Racher, business development director at Risk Decisions Ltd, agreed, adding that open dialogue between the sponsor and the contractor is absolutely essential.
“Unless you share project and risk management plans, you haven’t got the trust and you haven’t necessarily worked out which organisation is best placed to deal with each risk,” she said.
Share and share alike
For Viliam Kovac, vice president of global supply chain quality at Roche Diagnostics, it is critical for both parties to share the same vision.
“This results in better alignment and ensures the risk of potential conflicts or misunderstandings is reduced,” he explained. “If you reduce the complexity, you reduce the risk.”
“Increased use of outsourced clinical trial support has been supported by the expanding capabilities of contract research organisations (CROs).”
This certainly rings true from the CMO’s perspective. “Information sharing allows each party to understand and play their role in support of their mutual objectives,” said Zinselmeier. “Agreement on critical metrics throughout the lifecycle of a product, and collaboration, provides a mechanism to adapt to changing requirements.”
There is still a long way to go before this kind of openness becomes commonplace across the supply chain, particularly when it comes to working with CROs. “We need the culture to change to make it an expectation that risk will be a major discussion point,” said Bennett.
“One of the big problems I see in this network environment is the reluctance to be open about risk. Often sponsors don’t hear about things until it’s too late.”
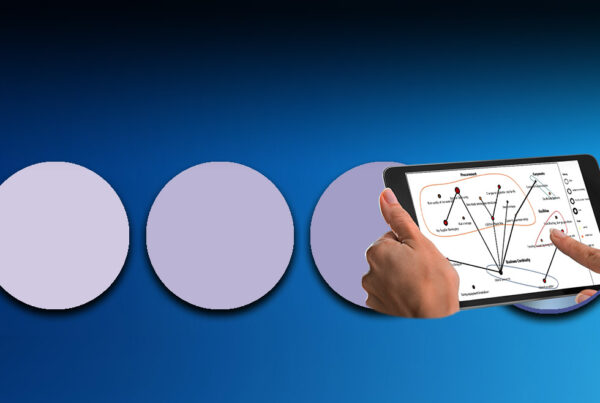
One way to improve this situation is by investing in the sophisticated risk management tools that are already used extensively across industries such as construction, defence and aerospace but remain relatively rare in the pharmaceutical industry.
They can greatly increase the impact and value of risk management efforts by, for example, allowing views of risks across the portfolio to be created with great ease and by giving sponsors and CROs consistent risk reports as projects progress. Roche Diagnostics is ahead of the trend and has been using its ‘Global Supply Chain and Risk Assessment Tool’ since 2004. This innovative tool identifies potential risk areas by using a systematic approach to check the combination of probability and possible impact.
At the end of this process a ‘risk number’ is provided and if this is higher than the company’s risk acceptance level, it is classified a ‘key risk’. “We perform a risk assessment every two years and the years in between are for implementing mitigation actions,” Kovac explained. “It’s a proactive approach and has a direct impact on all supply chain management.”
Opportunity knocks
Something that’s often forgotten is that where there are uncertainties and risks, there are also opportunities. “Clinical trials run early so infrequently that people aren’t in a position to take advantage of it when it does happen,” said Bennett.
“But if you’re not going to talk about risks in an open way, you certainly won’t be able to take advantage of the opportunities.”
Racher agreed. “There’s an awful lot to be gained on both sides from having a stronger risk management culture. Rather than it being something negative, pharmaceutical companies could really start to see the benefits of being open in their discussion and management of risk.”